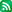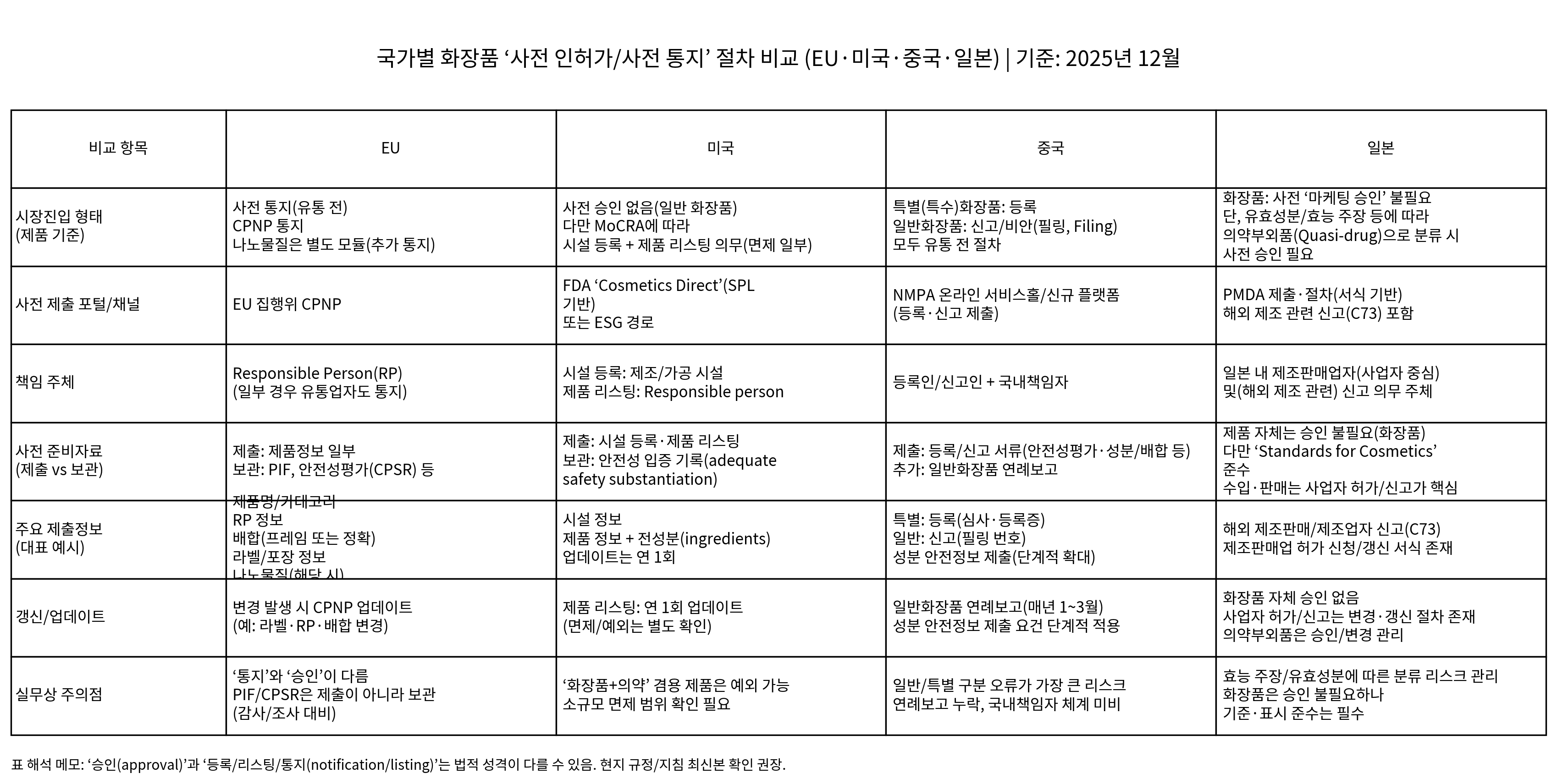
출처:
European Commission (Cosmetic Products Notification Portal, CPNP – Guidance on notification of cosmetic products, 최신 개정본)
European Commission
출처:
U.S. Food and Drug Administration (Modernization of Cosmetics Regulation Act of 2022 – Overview & Compliance Requirements, 2023–2024 업데이트)
U.S. Food and Drug Administration
출처:
U.S. Food and Drug Administration (Cosmetics Direct: Facility Registration & Product Listing Guidance, 2023–2024)
U.S. Food and Drug Administration
출처:
National Medical Products Administration (Provisions for Registration and Filing of Cosmetics, 시행 및 해설자료 포함, 최신본)
National Medical Products Administration
출처:
National Medical Products Administration (Cosmetic Registration & Filing Information Platform and Annual Reporting Requirements, 2023–2024)
National Medical Products Administration
출처:
Pharmaceuticals and Medical Devices Agency (Cosmetics Q&A – Marketing approval not required for cosmetics, 최신본)
Pharmaceuticals and Medical Devices Agency
출처:
Pharmaceuticals and Medical Devices Agency (Notification of Foreign Manufacturers / Marketing Authorization Holders for Cosmetics – Form C73, 최신본)
Pharmaceuticals and Medical Devices Agency
출처:
Pharmaceuticals and Medical Devices Agency (Standards for Cosmetics & Procedures for Quasi-drugs, 관련 가이드라인, 최신본)
Pharmaceuticals and Medical Devices Agency
와이에스엠경영컨설팅 윤수만